Ahmed® ClearPath™
Product Description:
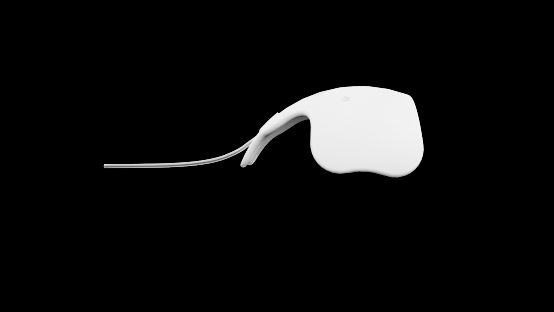
The new Ahmed® ClearPath™ is the latest non-valved drainage device designed to shunt aqueous in eyes suffering from refractory glaucoma.

Information
Videos
PDFs
Videos
The Ahmed® ClearPath™ offers:
EASE OF USE (Better Accessibility and Visibility)
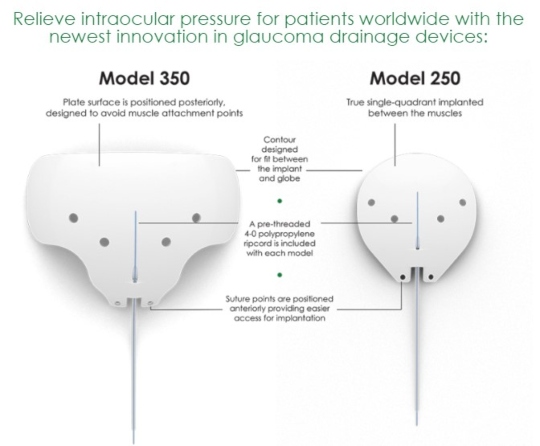
- Suture points of the plate are positioned anteriorly versus other models on the market, providing easier access for implantation.
CONVENIENCE (Efficient packaging)
- Both models include a preplaced 4 -inch 4-0 prolene ripcord suture and a pre-packaged 23G hypodermic needle for the convenience of the surgeon and OR staff during implantation.
SURGICAL EFFICIENCY (Better Fit and Placement)
- Model CP250 is a true single-quadrant device that fits between the muscles, eliminating the need to isolate them.
- Model CP350 has a winged design with the plate surface positioned posteriorly, designed to avoid muscle attachment points.
- Both models have a contour that is designed to naturally conform to the surface of the globe.
Product Features
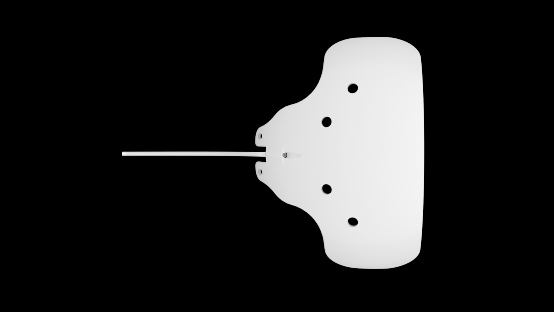
- The implant consists of a medical grade silicone tube secured near the anterior suture points of the medical grade silicone episcleral plate.
- Two models are available that cover surface areas of approximately 250 mm2 and 350 mm2.
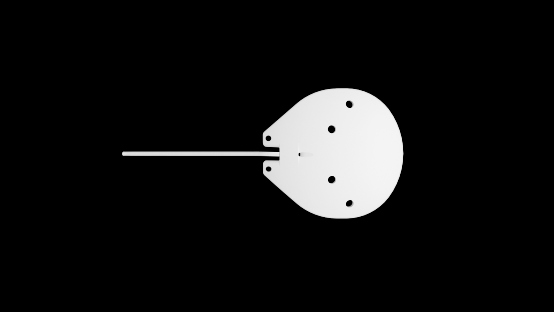
- The anterior suture points are located on the anterior side of the plate, flanking each side of the tubing track.
- The silicone plate is barium impregnated to increase ultrasound resolution and identification with CT scans, MRIs and plain skull films.
- The plate conforms to the shape of the globe at its equator and provides a surface from which fluid can be dispersed.
- The device is for single use only.
Related Products
Find More Individual Products with our Product Search Filter
Educational Insights

Drop it like it’s Hot: PRGF to the rescue for Ocular Surface Disease! Webinar
Brittany Yelle (Precision Cornea Center, Ottawa,...

PRN & BRUDER – Foundation of Dry Eye – Dr Kim Friedman
Dry eye is a very common condition, affecting...

The Foundations of Dry Eye Therapy – Dr Kim Friedman
Dry eye is a very common condition, affecting...

ENDORET: Ask the Expert with Dr Brittany Yelle
Plasma Rich in Growth Factors (PRGF) is a...